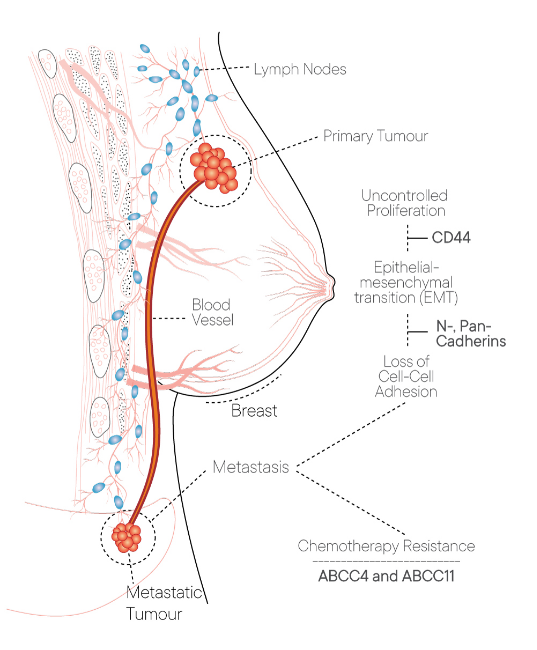
CanAssist Breast uses a proteomics- based method and a proprietary machine learning-based algorithm to to analyze a patented combination of protein biomarkers from the patient’s tumour to compute the risk of recurrence of cancer.
CanAssist Breast integrates IHC (immunohistochemistry) data and clinical parameters to calculate a risk score that classifies patients as ‘low-risk’ or ‘high-risk’ for breast cancer recurrence over five years. By integrating the tumour biology of the disease with time-tested clinico-pathological parameters we assist physicians in developing optimal treatment plans for cancer patients. The CanAssist Breast test result allows breast cancer patients who are classified as ‘low-risk’ to potentially avoid chemotherapy and its side effects.

